N2 EDUCATION
N2 works with the Collaborative Institutional Training Initiative (CITI) to provide high-quality, web-based, TransCelerate recognized Canadian instruction on a variety of research topics. These courses promote research with human participants to be conducted in a manner that is appropriate, safe, responsible, ethical and meets the guidelines, regulations and legislation in this country. The 8-courses in the CITI-Canada curriculum are composed of modules and quizzes; completion certificates are generated for successful learners.
In 2010 N2 partnered with the Collaborative Institutional Training Initiative (CITI), a not-for-profit entity at the University of Miami, which has since become part of the Biomedical Research Alliance of New York (BRANY).
N2 has the rights to add and modify all CITI-content to ensure it reflects and meets the educational needs of the Canadian research environment.
The CITI-Canada Program offers comprehensive courses on a variety of research related matters. The courses include a quiz after each module to test the learner’s knowledge. Following successful completion of the course, the learner is able to obtain a completion certificate. This can be used to demonstrate compliance with the regulatory requirements for education in the respective topics. The organization will be able to run aggregate reports on all staff in its organization to track completion of the respective courses.
N2 provides access to the CITI-Canada program for N2 member organizations. N2 does provide limited access for individuals for a nominal fee. If you are not a member but interested in accessing the program please contact us.
Course Descriptions
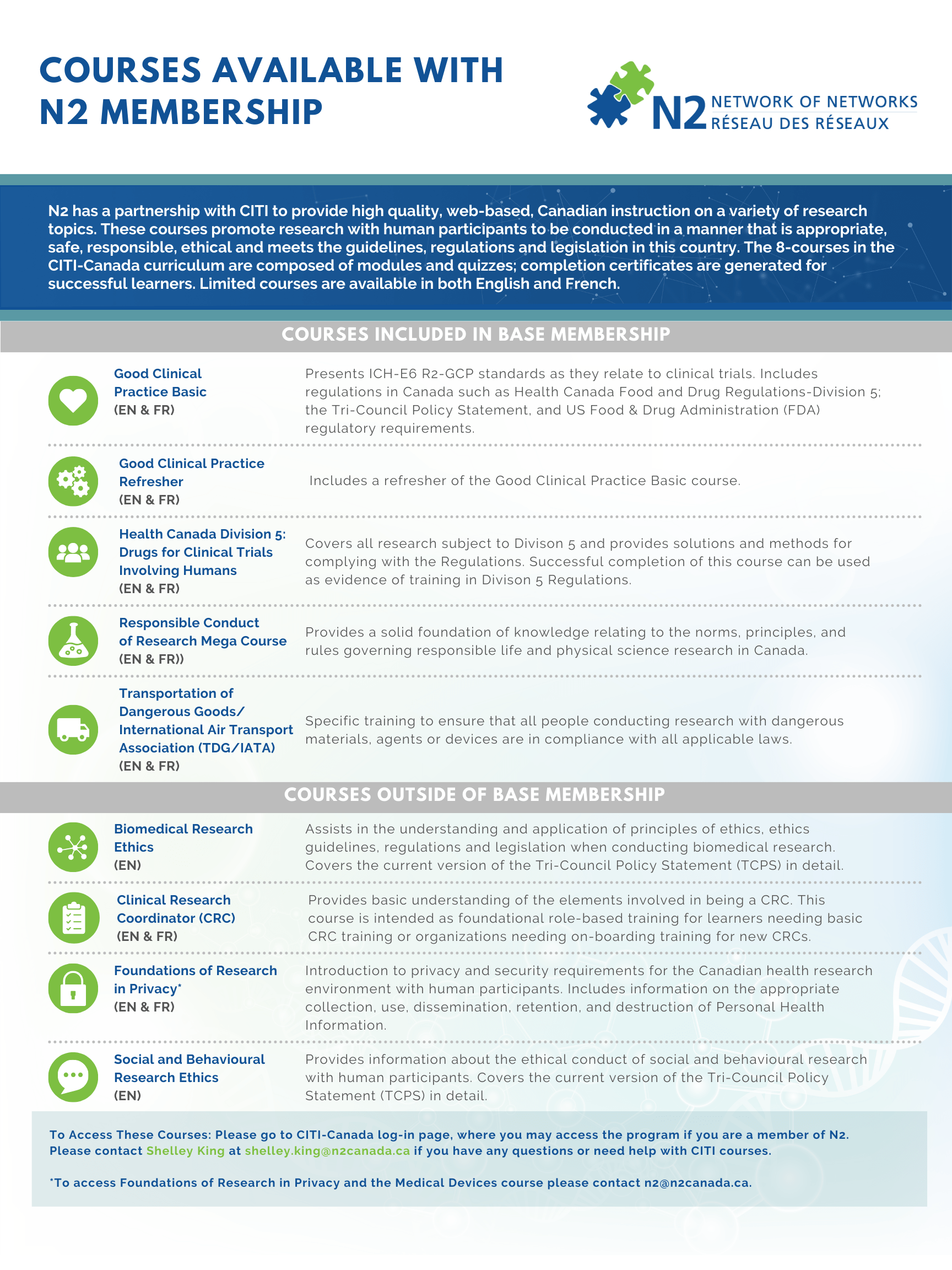
Courses Included in Base-Membership
Good Clinical Practice Basic (EN & FR)
This course is a 10-module program that discusses good clinical practice as it relates to clinical trials. The discussion is focused on the International Council for Harmonisation (ICH)-E6 (R2) guidelines and applicable regulations in Canada such as Health Canada-Division 5, and The Tri-Council Policy Statement 2.
CITI Canada’s GCP Courses were revised to reflect the updated ICH Harmonised Guideline Integrated Addendum to ICH E6(R1): Guideline for Good Clinical Practice E6(R2). GCP Courses – Version 2 – now specifically refer to the current ICH E6(R2) guideline and provide ICH E6(R2) training.
Both CITI Canada GCP courses meet the minimum criteria for ICH GCP Investigator Site Personnel Training identified by TransCelerate BioPharma as necessary to enable mutual recognition of GCP training among trial sponsors. Both the GCP Basic and Refresher courses are available in both French and English. The translation of the course was accomplished with financial support from Fond de Recherche Sante (FRQS) and with review support from Consortium de recherche en oncologie clinique du Québec (Q-CROC) . N2 is grateful for their contributions.
Good Clinical Practice (GCP)
- Version 1 – 28 Mar 2014
- Version 2 – 4 Apr 2018 (includes E6 R2 updates)
Who should take this course?
The course is suitable for persons proposing to conduct clinical trials primarily in Canada. “TransCelerate BioPharma” which represents a number of biopharmaceutical companies has recognized that the CITI Canada Good Clinical Practice course meets their criteria for ICH GCP training for investigator site personnel. This is significant in that N2 has worked with TransCelerate to assure that the high quality CITI Canada GCP course accessible to N2 members meets their standards. This means if you are working with a TransCelerate affiliated company you can assure them that your CITI Canada training has formal approval and recognition from TransCelerate. Meeting TransCelerate training criteria is the latest milestone in a trend that has seen many biopharmaceutical companies as well as research networks, hospitals and universities accept the CITI Canada GCP course as their gold standard.
Good Clinical Practice Refresher (EN & FR)
This course is a 7-module program that discusses good clinical practice as it relates to clinical trials. The discussion is focused on the International Council for Harmonisation (ICH)-E6 (R2) guidelines and applicable regulations in Canada such as Health Canada-Division 5, and The Tri-Council Policy Statement 2.
CITI Canada’s GCP Courses were revised to reflect the updated ICH Harmonised Guideline Integrated Addendum to ICH E6(R1): Guideline for Good Clinical Practice E6(R2). GCP Courses – Version 2 – now specifically refer to the current ICH E6(R2) guideline and provide ICH E6(R2) training.
Both CITI Canada GCP courses meet the minimum criteria for ICH GCP Investigator Site Personnel Training identified by TransCelerate BioPharma as necessary to enable mutual recognition of GCP training among trial sponsors. Both the GCP Basic and Refresher courses are available in both French and English. The translation of the course was accomplished with financial support from Fond de Recherche Sante (FRQS) and with review support from Consortium de recherche en oncologie clinique du Québec (Q-CROC) . N2 is grateful for their contributions.
CITI Canada GCP Refresher
- Version 1 – 19 Mar 2015
- Version 2 – 25 Jan 2017 (includes E6 R2 updates)
Who should take this course?
The course is suitable for persons proposing to conduct clinical trials primarily in Canada. “TransCelerate BioPharma” which represents a number of biopharmaceutical companies has recognized that the CITI Canada Good Clinical Practice course meets their criteria for ICH GCP training for investigator site personnel. This is significant in that N2 has worked with TransCelerate to assure that the high quality CITI Canada GCP course accessible to N2 members meets their standards. This means if you are working with a TransCelerate affiliated company you can assure them that your CITI Canada training has formal approval and recognition from TransCelerate. Meeting TransCelerate training criteria is the latest milestone in a trend that has seen many biopharmaceutical companies as well as research networks, hospitals and universities accept the CITI Canada GCP course as their gold standard.
Health Canada Division 5 - Drugs for Clinical Trials Involving Humans (EN & FR)
The Food and Drugs Act and the Food and Drug Regulations govern the sale and importation of drugs for use in human clinical trials in Canada. Specifically, Part C, Division 5 of the Regulations addresses drugs for clinical trials involving human participants. Health Canada tells us that the Qualified Investigator must ensure compliance with the Regulations and the International Conference on Harmonisation Good Clinical Practice (ICH-GCP E6) Guideline (incorporated in the Regulations) from every person involved in the conduct of the clinical trial at their site. To that end, there should be evidence of satisfactory training of personnel involved in these processes. The successful completion of this CITI-Canada course can be used as evidence of training in Division 5 Regulations. This “Division 5” course covers all research conducted under Division 5 Regulations and it provides practical solutions and methods for complying with the Regulations. This course is available in both French and English.
Responsible Conduct of Research Mega Course (EN & FR)
How should you conduct your research? What practices should you follow? Researchers are expected by the public and by their colleagues to follow many rules and commonly accepted practices. The Responsible Conduct of Research (RCR) courses provide the learner with a solid foundation of knowledge relating to the norms, principles and rules governing responsible research practice in Canada.
List of Modules for RCR Mega course:
- Introduction to RCR
- Ethics and the Responsible Researcher
- Publication Practices and Responsible Authorship
- Collaborative Research; Conflicts of Interest
- Data Management
- Mentoring
- Peer Review
- Writing with Integrity
- Research Involving Human Subjects
- Research Misconduct
- Using Animal Subjects in Research
Who should take this course?
These courses are suitable for researchers, students and other personnel from a variety of disciplines and fields.
Transportation of Dangerous Goods/ International Air Transport Association (TDG/IATA) (EN & FR)
Compliance with the Transportation of Dangerous Goods/ International Air Transport Regulations has become increasingly important. Specific training for investigators and staff on Transportation of Dangerous Goods/ International Air Transport Regulations is essential to ensure that all people conducting research using dangerous materials, agents or devices are in compliance with all applicable laws. The course consists of 6 modules and covers the following: Introduction; Classification; Marks of Safety; Packaging and Containment Systems, Marking and Labeling; Documentation; Emergency Response Assistance Plans (ERAPs) and Accidental Release Reporting.
NOTE: Please note that the TDG training requirements are quite non-specific and leave most of the responsibility to the employer. The employer determines whether an employee needs to be trained and what kind of training is necessary, and the employer is also the one who issues the certificate of training. This certificate has very specific requirements, and the one you will receive from CITI does not meet these requirements. It is therefore up to the site and users of the course to determine if additional education is required or beneficial. Many of our members use this course as a refresher.
The employer is responsible for providing the certificate of training per Transport Canada Regulations
Who should take this course?
The course is suitable for a broad range of audiences ranging from researchers working in labs that handle dangerous goods to clinical research staff or anyone involved in the transportation of dangerous goods and /or international air transport.
Courses Outside of Base-Membership
Biomedical Research Ethics (EN)
Biomedical Focus course consists of 9 modules and provides historic and current information on regulatory and ethical issues important to the conduct of research involving human participants.
The discussion is focused on introducing:
- Research Ethics Guidelines and regulations
- Research Ethics Boards and Ethics review process
- Harms and Benefits
- Process of Free and Informed Consent
- Privacy and Confidentiality
- Conflict of Interest
- Special Research Design Issues
Who should take this course?
The basic course modules are suitable for all persons involved in biomedical research studies involving human participants.
Clinical Research Coordinator (CRC) (EN & FR)
The Clinical Research Coordinator (CRC) Course was developed for CITI-Canada in order to provide learners with a basic understanding of the elements involved in being a CRC. This course is intended as foundational role-based training for learners needing basic CRC training or organizations needing on-boarding training for new CRCs. It offers learners a foundation that expands beyond but is directly connected to the Biomedical Research Ethics course and the Good Clinical Practice (GCP) training offered by N2/CITI-Canada. We recommend you complete the N2/CITI-Canada course on “Biomedical Research Ethics” which provides an excellent background on research ethics involving human participants, prior to completing this course.
List of Modules for CRC course:
- Overview
- Planning Research
- Funding
- Working with the Research Ethics Board (REB)
- Protocol Review and Approvals
- Principal Investigator (PI) Responsibilities
- Clinical Research Coordinator (CRC) Responsibilities
- Sponsor Responsibilities
- Informed Consent
- Site Management
- CRC Resources
- Overview of the Clinical Trial Agreement (CTA)
- Coordinating U.S. Regulated Studies – What to Consider?
Who should take this course?
The content of this course is intended for CRCs, investigators, and other clinical research professionals. It is also useful to those pursuing a career in clinical research. This course is recommended for clinical research staff who have 1-2 years’ experience in clinical research. This course is available in both French and English.
Foundations of Research in Privacy (EN & FR)
Research organizations are increasingly faced with challenges surrounding the management and security of the information they collect and use during the course of conducting research. N2 has developed this introductory course on privacy and security for personal information (PI) and personal health information (PHI) in the research environment. The course is designed to equip learners with an understanding of the privacy legislation around how personal information may be collected, used, shared, accessed and disposed; the fundamentals of records and information management; and leading practices for handling a privacy breach. This course targets individuals who administer, review, or conduct health research that involves collecting health data, such as during clinical trials.
Who should take this course?
This course is anyone who administers, reviews or conducts health research (which is research that involves the collection of health data such as occurs in – but is not limited to – clinical research).
Please contact n2@n2canada.ca for information on how to access this course.
Social and Behavioural Research Ethics (EN)
This course is an introduction to a variety of ethics issues that are important to consider when conducting social and behavioural research with human participants. It covers the current version of the Tri-Council Policy Statement (TCPS) in greater detail than the tutorial offered by the Panel on Research Ethics, with more information that is specifically aimed at a social and behavioural research audience.
List of Modules for Social and Behavioural Research Ethics course:
- Introduction
- History and Ethical Principles
- Defining Research with Human Participants
- The Tri-Council Policy Statement
- Assessing Risk in Social and Behavioural Sciences
- Informed Consent
- Privacy and Confidentiality
- Research with Children
- Research Participants in Vulnerable Circumstances
- Internet Research
- Conflicts of Interest
Who should take this course?
This course is suitable for all persons involved in social and behavioural research with human participants, such as healthcare providers, academics, researchers, research personnel, REB members, administrators and research coordinators.
Independent Learner Registration